Chronic pain
Pain is an important sensory modality that provides information about the risk or occurrence of tissue damage. People that are not able to experience pain, due to a genetic defect, suffer from chronic injuries or infections and they have a reduced life expectancy. An increased pain sensation has a protective role inducing behavioural changes that protects the individual from further injuries and assists at wound repair. However, a chronic pain condition, which may arise as a consequence of tissue inflammation or damage to sensory nerves, is a pathological condition that seriously decreases the quality of life. Unfortunately, chronic pain is difficult to treat. We therefore investigate the molecular and cellular mechanisms underlying such chronic conditions in the hope that our research may eventually contribute to a better therapy.
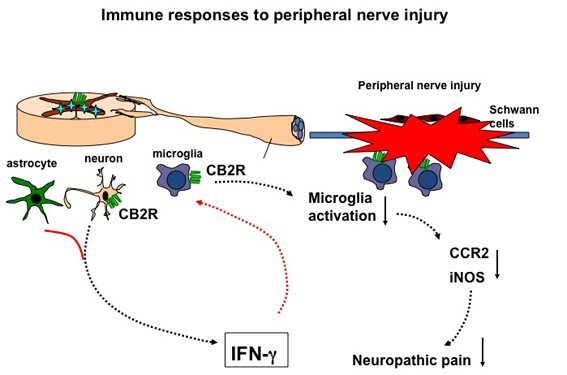
In the focus of our interest is the role of the endocannabinoid system, especially CB2 receptors in neuroinflammatory and neuropathic pain conditions. The interest in the medical use of cannabinoid ligands has greatly increased in the last few years, because both cannabinoid receptors (CB1 and CB2) (Matsuda et al., 1990; Munro et al., 1993) were shown to modulate neuropathic pain. Indeed, cannabis derivatives have been used for centuries as analgesic compounds, and cannabinoid agonists have been reported to be effective in several animal models of acute and chronic pain (Pertwee, 2001). However, the psychoactive effects of these agonists could represent a serious limitation for their clinical use, whereby CB1 receptors (mainly located in the central nervous system (Tsou et al., 1998)) seem to be selectively involved here (Ledent et al., 1999; Zimmer et al., 1999). The identification of CB2 receptors in glial cells (Zhang et al., 2003; Beltramo et al., 2006) has opened new therapeutic approaches for these ligands, because CB2 receptor activation induces analgesic effects in several animal models. The use of selective CB2 receptor agonists therefore represents a promising approach for the development of analgesic and anti-inflammatory therapies (Racz et al., 08).
Our previous research has indicated that CB2 receptors regulate inflammatory responses in the central nervous system after peripheral nerve injury by modulating the activity of microglia cells. Microglia activity is enhanced under neuropathic pain conditions in mice with a genetic deletion of the CB2 receptor encoding Cnr2 gene and also enhanced in transgenic animals overexpressing this receptor. We are currently investigating the molecular mechanisms that contribute to the enhanced microglia response and address the possible involvement of other cells in the neuropathic pain phenotype of CB2 knockout mice.
Following the pain research of our laboratory, we further established a comprehensive pain phenotyping platform in collaboration with the German Mouse Clinic (GMC), which is a phenotyping centre with open access to the scientific community. Here, mutant mouse lines are systematically characterized using standardized protocols. We have established sensitive screening methods that can reliably detect a broad range of changes in pain behavior.
This research area is supported by Bundesministerium für Bildung und Forschung (NGFN plus).
References:
Beltramo M, Bernardini N, Bertorelli R, Campanella M, Nicolussi E, Fredduzzi S, Reggiani A. CB2 receptor-mediated antihyperalgesia: possible direct involvement of neural mechanisms. Eur J Neurosci 2006;23(6):1530-1538.
Ledent C, Valverde O, Cossu G, Petitet F, Aubert JF, Beslot F, Bohme GA, Imperato A, Pedrazzini T, Roques BP, Vassart G, Fratta W, Parmentier M. Unresponsiveness to cannabinoids and reduced addictive effects of opiates in CB1 receptor knockout mice. Science 1999;283(5400):401-404.
Matsuda LA, Lolait SJ, Brownstein MJ, Young AC, Bonner TI. Structure of a cannabinoid receptor and functional expression of the cloned cDNA. Nature 1990;346(6284):561-564.
Munro S, Thomas KL, Abu-Shaar M. Molecular characterization of a peripheral receptor for cannabinoids. Nature 1993;365(6441):61-65.
Pertwee RG. Cannabinoid receptors and pain. Prog Neurobiol 2001;63(5):569-611.
Racz I, Nadal X, Alferink J, Banos JE, Rehnelt J, Martin M, Pintado B, Gutierrez-Adan A, Sanguino E, Manzanares J, Zimmer A, Maldonado R. Crucial role of CB(2) cannabinoid receptor in the regulation of central immune responses during neuropathic pain. J Neurosci 2008;28(46):12125-12135.
Zhang J, Hoffert C, Vu HK, Groblewski T, Ahmad S, O'Donnell D. Induction of CB2 receptor expression in the rat spinal cord of neuropathic but not inflammatory chronic pain models. Eur J Neurosci 2003;17(12):2750-2754.
Zimmer A, Zimmer AM, Hohmann AG, Herkenham M, Bonner TI. Increased mortality, hypoactivity, and hypoalgesia in cannabinoid CB1 receptor knockout mice. Proc Natl Acad Sci U S A 1999;96(10):5780-5785.





